Carbon 14 Dating Calculator

Ex: Exponential Model - Determine Age Using Carbon-14 Given Half Life
Radiocarbon Dating Calculations (Question 1)
Science > Biology library > And now, we can get our calculator out and just solve for what this time is. It uses the naturally occurring radioisotope carbon-14 ( 14C) to estimate the age of carbon-bearing materials up to about 58,000 to 62,000 years old. Oct 20, 2023 · By looking at the ratio of carbon-12 to carbon-14 in the sample and comparing it to the ratio in a living organism, it is possible to determine the age of a formerly living thing fairly precisely.
4.5.8 Exponential Decay: Carbon-14 dating
N (t) is the number of carbon-14 atoms left in the . To find the percent of Carbon 14 remaining after a given number of years, type in the number of years and click on Calculate. You have estimated that your sample presently contains about 400 units of carbon-14 (N t = 400), or about 40% of the . While 12 C is the most abundant carbon isotope, there is a close to constant ratio of 12 C to 14 C in .

The parameters used for the corrections have been obtained through precise radiocarbon dating of thousands of samples . Taking Noah’s flood into consideration changes carbon 14 dates. I.
Carbon Dating - Example of exponential decay
This radiocarbon dating calculator uses the carbon 14 dating technique to determine the age of archaeological artifacts from the percentage of carbon-14 ( 14 C) left in it. T designation SRM 4990 B and is termed HOx1. In this video, I want to go through a concrete example.


Carbon-14 Biblical date calculator.

(N 0 = 1,000).


T (National Institute of Standards and Technology; Gaithersburg, Maryland, USA) Oxalic Acid I (C2H2O4). .
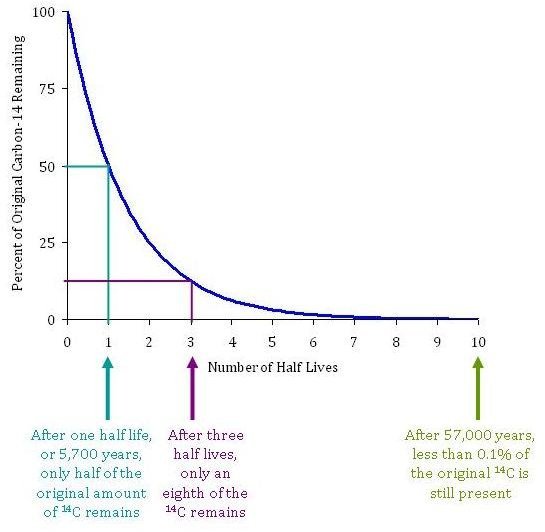

Carbon-14 is an isotope of carbon that is produced when solar neutrinos strike 14 N 14 N particles within the atmosphere. Let us determine the age of a wooden object found in an archaeological dig using our online radiocarbon dating calculator.

Info about c14 .

The half-life of carbon 14 is 5,730 … See more. When an organism dies, its carbon-14 decays back into nitrogen-14 at a known rate, called a half-life (5,730 years).

Calibrations of radiocarbon age determinations are applied to convert the Conventional Radiocarbon Age (BP age corrected for isotopic fractionation using a δ13C value) to calendar years. Carbon 14 is a common form of carbon which decays over time. Its presence in organic materials is the basis of the radiocarbon dating method pioneered by Willard Libby and colleagues (1949) to date archaeological, geological and hydrogeological samples.

The Carbon-14 Cycle.

Carbon-14 forms in the atmosphere through cosmic rays interacting with nitrogen-14, creating a small fraction of carbon-14 in living organisms.

Carbon-14, C-14, 14 C or radiocarbon, is a radioactive isotope of carbon with an atomic nucleus containing 6 protons and 8 neutrons. You can get an idea of the relationship between C14 and age at the Carbon Dating calculator page .

(0)
No comments yet. Be the first!